Adhesive plasters
 Registration Certificate for fixing adhesive plasters
Registration Certificate for fixing adhesive plasters
 Registration Certificate for medical adhesive plasters
Registration Certificate for medical adhesive plasters
 Registration Certificate for sets of bactericidal adhesive plasters
Registration Certificate for sets of bactericidal adhesive plasters
 Instructions for use of medical bactericidal adhesive plasters in individual packages
Instructions for use of medical bactericidal adhesive plasters in individual packages
 Instructions for use of sets of bactericidal adhesive plasters
Instructions for use of sets of bactericidal adhesive plasters
 Instructions for use of fixing adhesive plasters
Instructions for use of fixing adhesive plasters
 Instructions for use of medical non-woven adhesive plasters
Instructions for use of medical non-woven adhesive plasters
 Instructions for use of bactericidal polymer adhesive plasters
Instructions for use of bactericidal polymer adhesive plasters
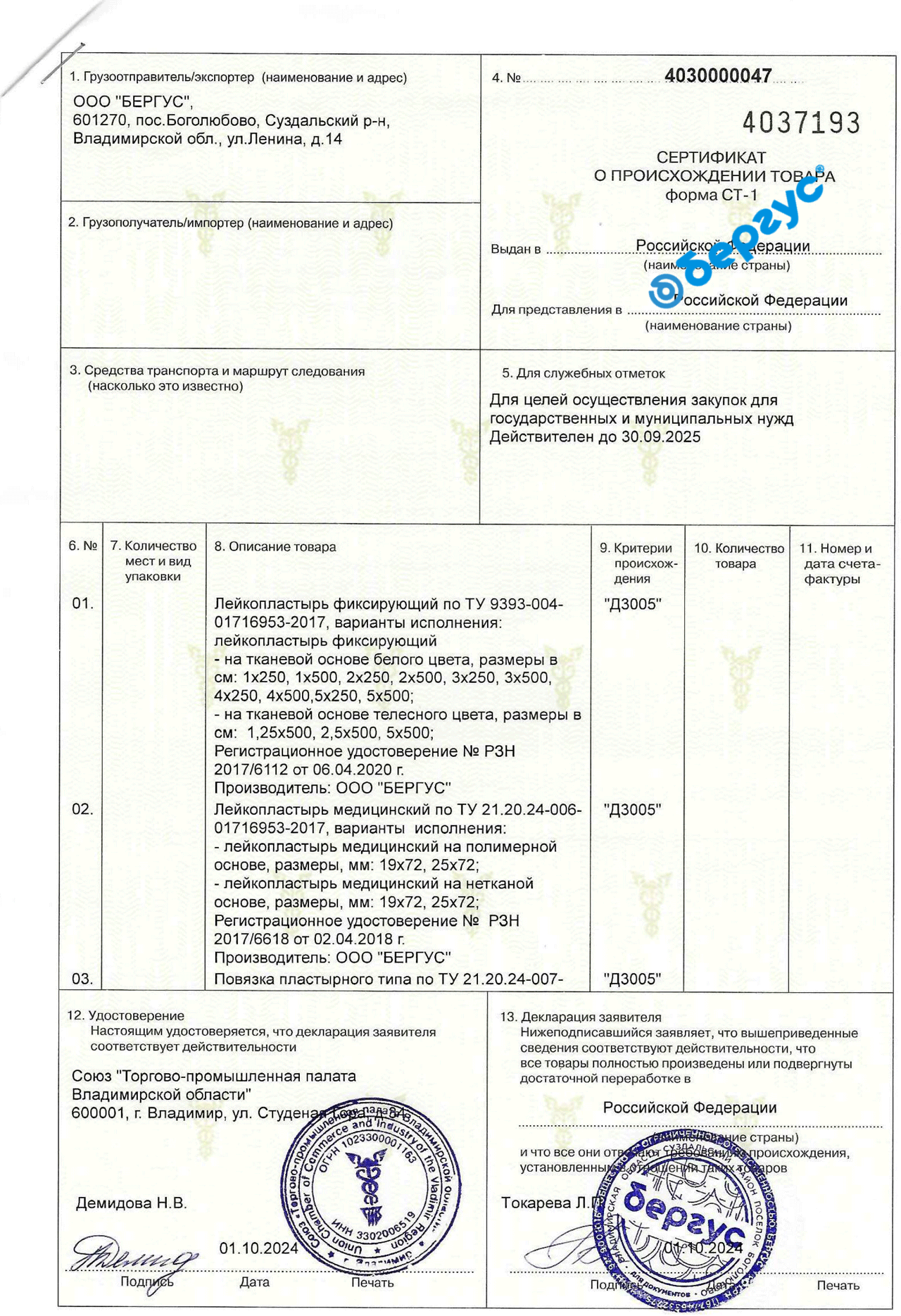 Certificate of Origin of goods CT-1 (adhesive plasters)
Certificate of Origin of goods CT-1 (adhesive plasters)
 Registration Certificate No RZN2017-6421 dated 18-04-2022
Registration Certificate No RZN2017-6421 dated 18-04-2022
This page presents registration documents for adhesive plasters produced by the company "Bergus".
The main documents are the registration certificate and the declaration of conformity. They confirm that the product has passed all necessary tests and can be sold and used in Russia.
Please note that some manufacturers' patches do not have a registration certificate and cannot be used for medical purposes or participate in tenders for healthcare facilities.
Adhesive plasters from the Bergus company are medical devices.
In addition, they have a certificate "ST-1". It is issued by the Chamber of Commerce and Industry of the Vladimir Region and confirms the Russian origin of the goods. "ST-1" provides additional advantages when participating in state and municipal tenders.
Documents for adhesive plasters
Due to the fact that our company is constantly expanding its range, there may be situations when not all documents for adhesive plasters are presented on the site. In this case, you can contact your personal manager for the necessary documentation.
If necessary, wholesale customers can also request a quality certificate for a separate batch of products, as well as certificates for trademarks.
The documents presented for adhesive plasters also apply to products manufactured by Bergus under the trademarks of partners (private labels).


